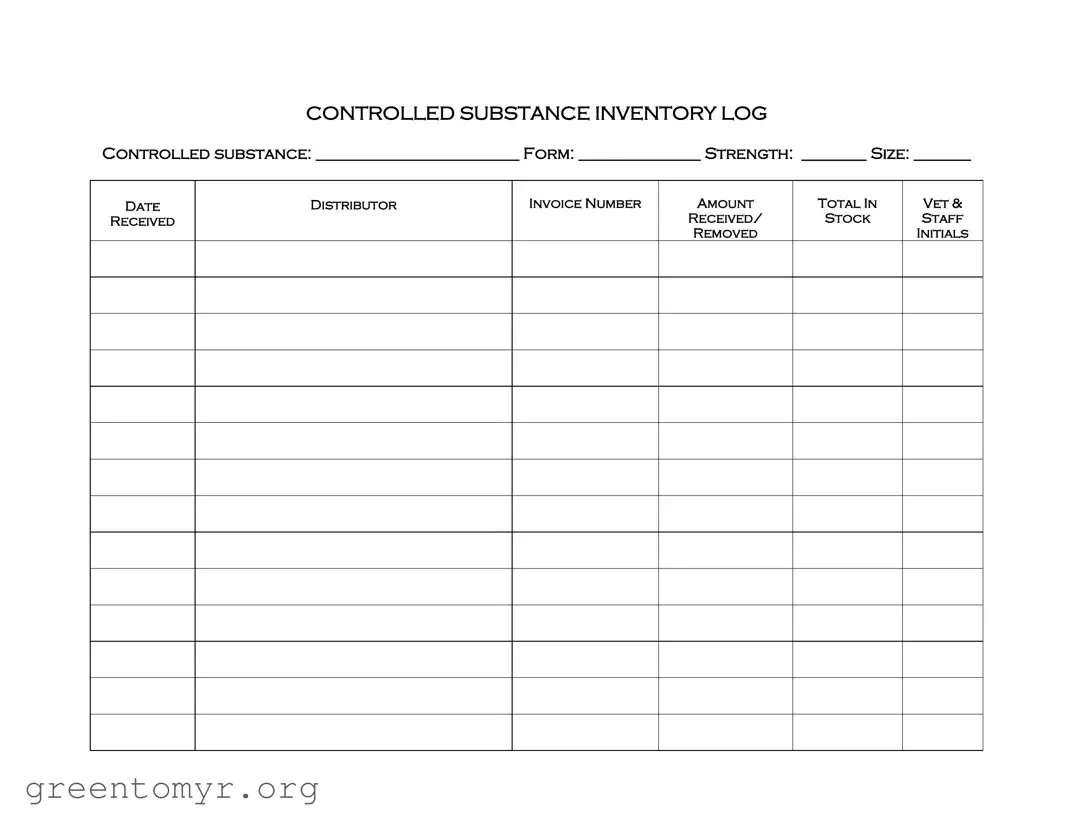
Filling out the Controlled Drug Log form accurately is crucial for compliance and accountability. However, many individuals make common mistakes that can lead to issues down the line. One frequent error is failing to include all required information. Each section of the form must be completed, including the controlled substance name, form, strength, size, and relevant dates. Omitting any of these details can create confusion and lead to discrepancies in inventory records.
Another mistake is inaccurate data entry. When recording information such as the invoice number or amount received, it's essential to double-check for typos or miscalculations. Errors in these fields can result in improper tracking of controlled substances, which may have legal repercussions.
Some individuals overlook the importance of initials and signatures. Each entry must be authenticated by the staff responsible for the transaction. Neglecting to initial the log can lead to questions about who handled the substances, making it difficult to trace accountability if an issue arises.
Additionally, not updating the log in real-time can cause significant problems. Waiting to fill out the log until later can lead to forgotten details or inaccurate records. It's best practice to update the log immediately after a transaction to maintain accurate inventory records.
Another common mistake is failing to maintain a consistent format. The log should follow a clear and organized structure. For instance, dates should be recorded in the same format throughout the document. Inconsistencies can lead to confusion and make it difficult to analyze the data later.
Some users also forget to cross-reference with other records. The Controlled Drug Log should align with other inventory records and invoices. Discrepancies between these documents can raise red flags during audits, so ensuring they match is essential.
Finally, neglecting to review the log periodically can lead to oversights. Regularly reviewing the log helps identify any errors or inconsistencies early on. This proactive approach can prevent larger issues from developing and ensures compliance with regulations.